Protector?? o Bloqueador?? solar sabes cual es la diferencia ---> síguenos para más contenido en FACEBOOK https://www.facebook.com/Three-Points-Make… | Makeup, Vida

La OCU responde: qué SPF debe tener una crema solar y qué significa realmente el factor de protección | Moda y belleza

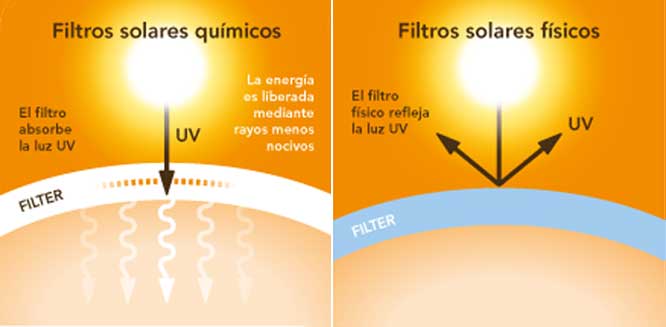
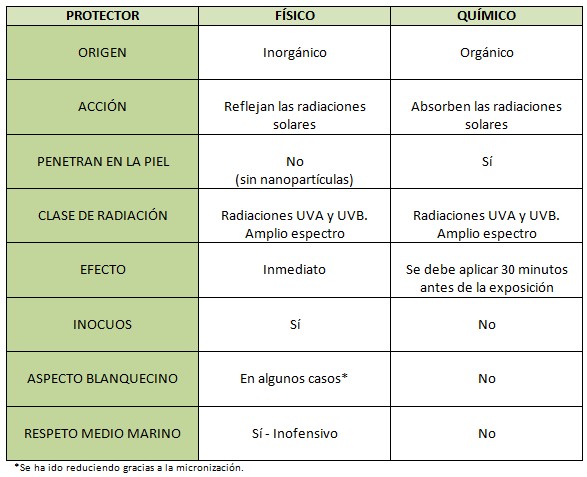
Shelo nabel Gonzalez'D - Protector, pantalla o bloqueador solar? ☀️ Que diferencia hay entre cada uno de ellos? 🤔 👉 Protector Solar: Filtra los rayos ultravioleta del sol y los mantiene apagados

☀ Diferencia entre PROTECTOR solar, BLOQUEADOR solar, PANTALLA solar y FILTRO solar 👀 Cremas SOLARES - YouTube